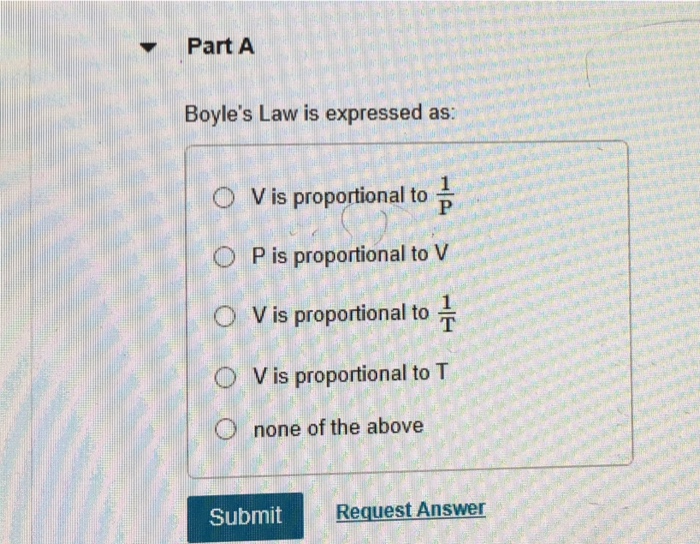
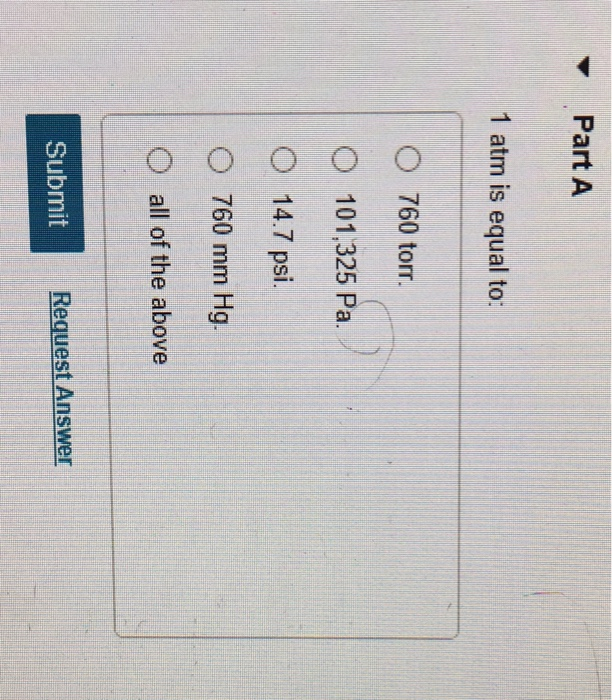
OneClass: Which of the following is not equivalent to 1 atm pressure? A) 10 cm Hg B) 14.7 lb/in^2 C) ...

Gas Pressure Unit Conversions - torr to atm, psi to atm, atm to mm Hg, kpa to mm Hg, psi to torr - YouTube

SOLVED: 1. Sl unit for pressure is A) atm: B) mm Hg (mercury) C) N/m D) Pascal E) Kg/m? 2. Sl unit for pressure is A) N/m B) kg/m? C) N/mz D)

SOLVED: 21. Calculate the value of R, the gas constant; when 3 moles of He gas is placed under a pressure of 1 atm at OC with a volume equal to 67.2







:max_bytes(150000):strip_icc()/GettyImages-124564231-58b5c7d75f9b586046caddb9.jpg)