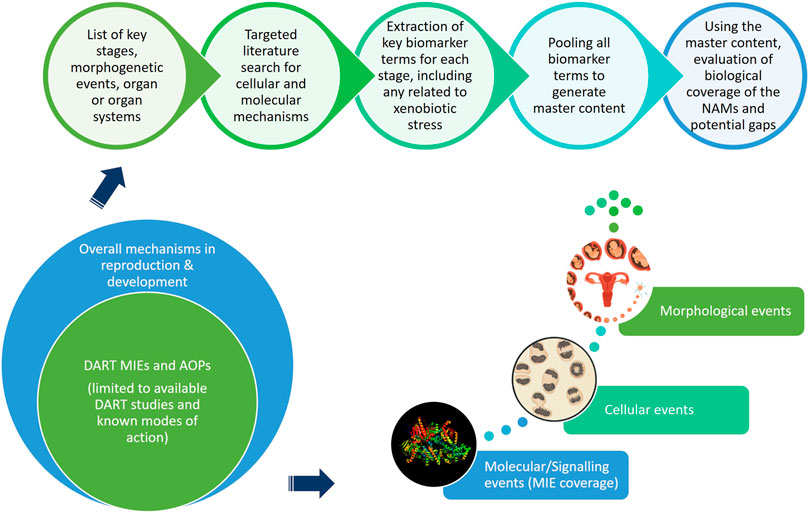
Considerations for Assessment of Reproductive and Developmental Toxicity of Oligonucleotide-Based Therapeutics | Nucleic Acid Therapeutics

FDA Guidelines For Developmental and Reproductive Toxicology (DART) Studies for Small Molecules | Leaders in Pharmaceutical Business Intelligence (LPBI) Group

Immunoaffinity nanogold coupled with direct analysis in real time (DART) mass spectrometry for analytical toxicology - Analytical Methods (RSC Publishing)

Table 2 from Subject : Critique of 15 DART Studies for Deltamethrin ( Re : OEHHA ' s Data Call-in ) | Semantic Scholar
DART-02: Prenatal Development Studies of 4-Methylcyclohexanemethanol (CASRN 34885-03-5) in Sprague Dawley (Hsd:Sprague Dawley SD

Characterization of bone abnormalities from micro-CT images for evaluating drug toxicity in developmental and reproductive toxicology (DART) studies | Semantic Scholar
Pharmaceutical toxicology: Designing studies to reduce animal use, while maximizing human translation

FDA Guidelines For Developmental and Reproductive Toxicology (DART) Studies for Small Molecules | Leaders in Pharmaceutical Business Intelligence (LPBI) Group