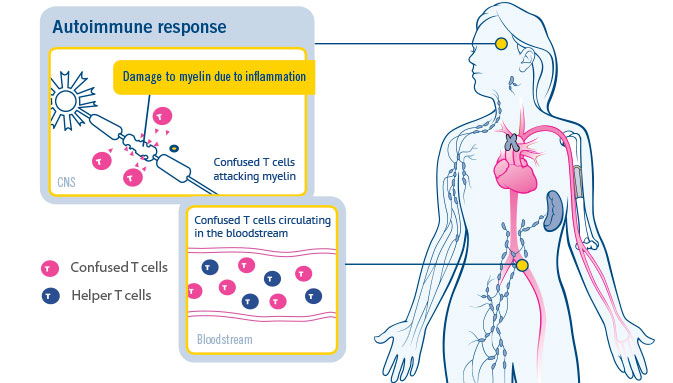
Teva Pharmaceutical Industries Ltd. - Relapsing remitting #MS (RRMS) is the most common form of multiple sclerosis. Learn more about MS in our full infographic https://bit.ly/3fP8jvu #MultipleSclerosis #MSConnections #MS #WorldMSDay | Facebook

Teva Pharmaceutical Industries' COPAXONE 40 mg/mL Three-times-a-week Approved in Russia for Relapsing-Remitting Multiple Sclerosis

TEVA stock as EU cites antitrust concerns over multiple sclerosis therapy (NYSE:TEVA) | Seeking Alpha